
More than 3.1 million bottles of eye drops sold at major nationwide retailers are being recalled because they may not be sterile, the FDA has said in a notice.
The eye drops have been recalled by the California-based manufacturer, K.C. Pharmaceuticals, which sells products under several brand names, including Dry Eye Relief Eye Drops and Sterile Eye Drops Redness Lubricant.
The recall involves eight brands manufactured by K.C. Pharmaceuticals, with the largest lot involving more than 1 million bottles sold under the Dry Eye Relief Eye Drops brand at stores including Rite Aid, H-E-B and Harris Teeter.
The FDA said the eye drops were distributed by companies such as CVS, Kroger, Walgreens, Dollar General, H-E-B, Military Exchanges and others.
The recall was classified as Class II by the FDA, which describes products that may cause “temporary or medically reversible adverse health consequences or where the probability of serious adverse health consequences is remote.”
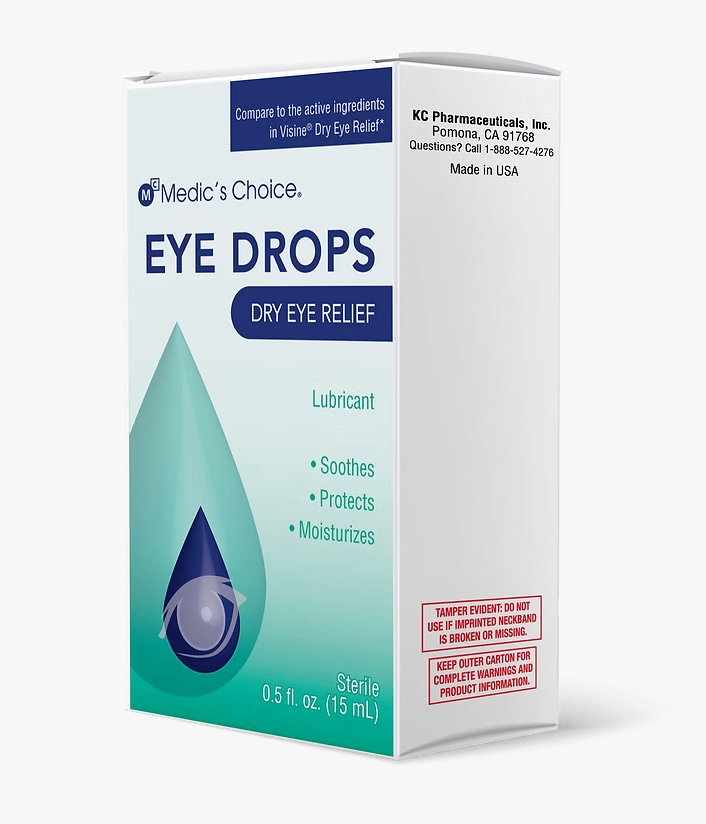
Many of them can be identified through their Universal Product Code or National Drug Code numbers, which are typically found near the barcode on the bottle.
The recalled items are below:
- 182,000 bottles of Sterile Eye Drops AC. Sold at stores including Walgreens, Meijer and H-E-B.
- 303,216 bottles of Eye Drops Advanced Relief. Sold at stores including Walgreens, Kroger and CVS.
- 1,023,096 bottles of Dry Eye Relief Eye Drops. Sold at stores including Rite Aid, H-E-B, Meijer and Harris Teeter, as well as military exchanges.
- 245,184 bottles of Ultra Lubricating Eye Drops. Sold at stores including Leader and Harris Teeter.
- 378,144 bottles of Sterile Eye Drops Original Formula. Sold at stores including Walgreens, Kroger, CVS, H-E-B, Harris Teeter, Dollar General, Circle K and Leader.
- 315,144 bottles of Sterile Eye Drops Redness Lubricant. Sold at stores including Walgreens, Rite Aid, Leader and Equaline, as well as workplace distributor Cintas.
- 74,016 bottles of Sterile Eye Drops Soothing Tears. Sold at Walgreens and through Rugby Laboratories.
- 589,848 bottles of Artificial Tears Sterile Lubricant Eye Drops. Sold at stores including Kroger, Publix, Leader and TopCare, as well as through Good Neighbor Pharmacy and Good Sense.
The recall was initiated on March 3 and then classified on March 31.
While there was no press release issued for this recall, it is typically recommended that consumers discard recalled products or return them to the place of purchase for a refund. The Independent has contacted K.C. Pharmaceuticals for comment.
You can find more information on the recalled products, including lot codes and UPC codes, in the FDA’s report.




















